Tue, 14 March 2023
In this first ever CE episode, we discuss the A-Zs of continuous glucose monitors (CGMs). In specific, our learning objective for the CE are: -
Describe commonly available types of continuous glucose monitors (CGMs) in the US market and the features and capabilities of these devices. -
Summarize the evidence and guideline recommendations for use of CGMs in the management of diabetes. -
Identify the role of the pharmacist in the selection of CGMs and provision of education to patients and providers. -
Interpret the ambulatory glucose profile (CGM data output) and recommend changes in antihyperglycemic regimen for a patient. ACPE-Accredited Pharmacist CE (1.0 hrs) To obtain CE credit for a $5 fee, visit the following link: https://rfums.wufoo.com/forms/z1qzh5vf0ggr832/. Once payment is successful, you will be redirected to our CE partner (CE Impact) to complete an evaluation and to earn 1.0 hour of CE credit. CE is available for 12 months after episode publication. Key Concepts - There are two main types of stand-alone personal CGMs available in the US market – real-time (rtCGM) and intermittently scanning (isCGM). [1] These CGMs vary in their features such as sensor wear time, sensor warm up time, sensor application site, reader availability, approved age for use, fingerstick calibration, non-adjunctive FDA labeling, interconnectability with other technology such as insulin pumps, and drug interactions – these variabilities can be used in decision-making when selecting an appropriate CGM for a patient. [2-7]
- Based on the evidence for use, both types of CGMs (real-time and intermittently scanning) are recommended in patients with Type 1 and Type 2 diabetes who are on multiple-daily insulin or continuous insulin infusion (pump), patients with Type 2 diabetes on basal insulin therapy, and as adjunct use in patients with diabetes who are pregnant. The strength of recommendations in general is stronger for real-time CGMs than for intermittently scanning CGMs. [1,11] These recommendations are supported by the evidence that CGMs can help improve glucose control, reduce risk of hypoglycemia, diabetes-related hospitalizations, and patient/caregiver satisfaction.
- Pharmacists play an integral role in education, on-going support, data interpretation, and resulting disease management in patients who qualify for CGM use and providers who care for patients with diabetes. [14]
- The ambulatory glucose profile is a standardized data output that informs understanding of glucose trends. [15] The recommended goal for most patients is to maintain a glucose range between 70-180 mg/dL with at least 70% of time spent in this range with variability coefficient of no more than 36%. [1,11,15]
Supplemental Content Comparison of rtCGM and isCGM devices 
"Mary's" Example AGP Report (adapted from Battelino et al.)  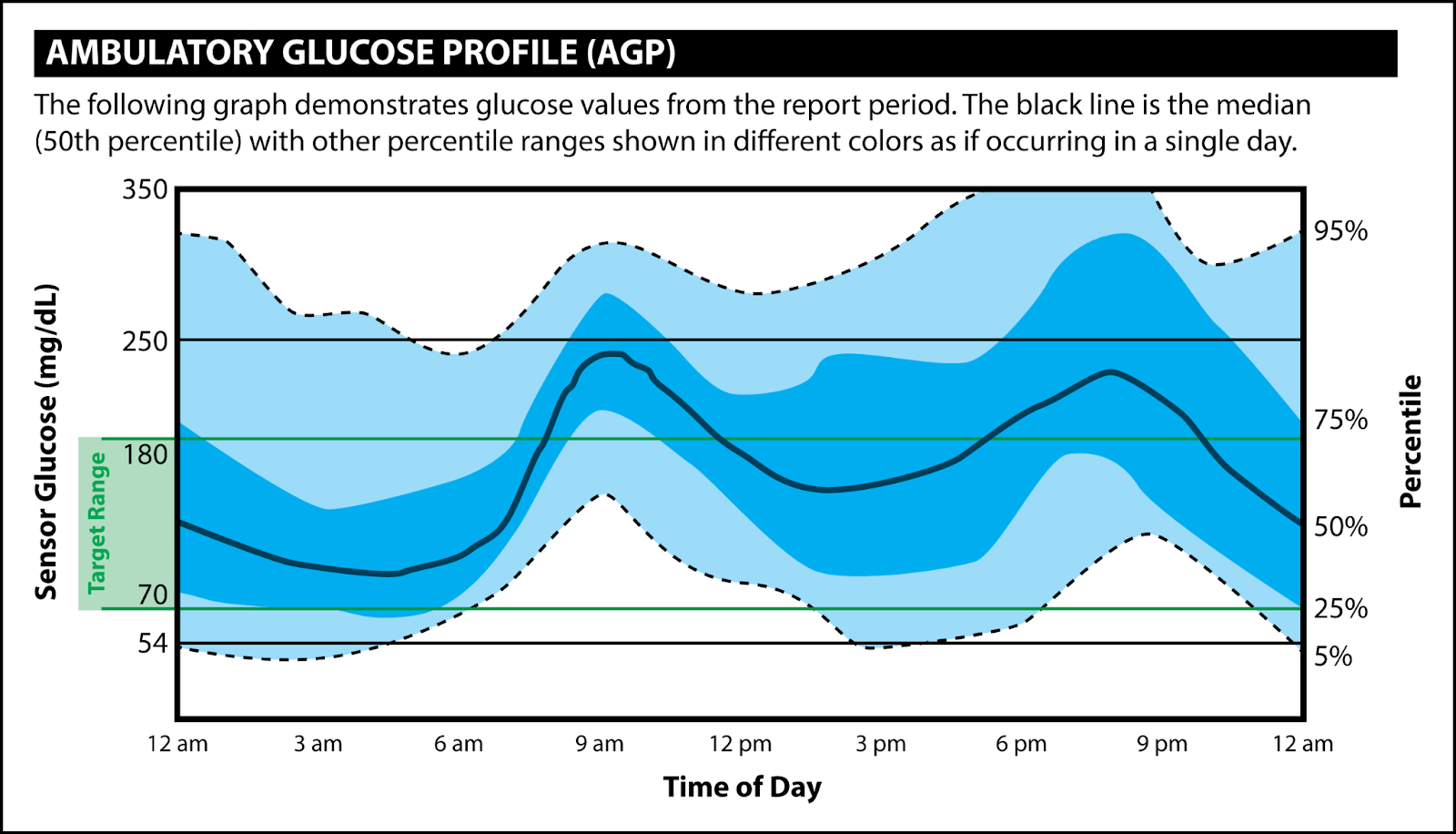
References - ElSayed NA, Aleppo G, Aroda VR, et al. American Diabetes Association. Chapter 7. Diabetes technology: Standards of medical care in diabetes - 2023. Diabetes Care. 2023;46(suppl 1):S111-S127.
- Dexcom G6 User Guide. Dexcom, Inc. 2020. Accessed February 20, 2023. https://s3-us-west-2.amazonaws.com/dexcompdf/G6-CGM-Users-Guide.pdf.
- Dexcom G7 User Guide. Dexcom, Inc. 2022. Accessed February 20, 2023. https://dexcompdf.s3.us-west-2.amazonaws.com/en-us/G7-CGM-Users-Guide.pdf#page=12
- Guardian Connect System User Guide. Medtronic MiniMed. 2020. Accessed February 20, 2023. https://www.medtronicdiabetes.com/sites/default/files/library/download-library/user-guides/Guardian-Connect-System-User-Guide.pdf.
- Eversense E3 User Guide. Sensionics, Inc. 2022. Accessed February 20, 2023. https://www.eversensediabetes.com/wp-content/uploads/LBL-4002-01-001-Rev-F_Eversense-E3-User-Guide_mgdL_R1_web.pdf
- FreeStyle Libre 3 User’s Manual. Abbott Diabetes Care Inc. 2022. Accessed February 20, 2023. https://freestyleserver.com/Payloads/IFU/2022/q2/ART44140-002_rev-A.pdf
- FreeStyle Libre 2 User’s Manual. Abbott Diabetes Care Inc. 2020. Accessed February 20, 2023. https://freestyleserver.com/Payloads/IFU/2020/q2/ART40703-001_rev-D-Web.pdf.
- Products. American Diabetes Association. Accessed February 20, 2023. https://consumerguide.diabetes.org/
- Wood A, O'Neal D, Furler J, Ekinci EI. Continuous glucose monitoring: a review of the evidence, opportunities for future use and ongoing challenges. Intern Med J. 2018 May;48(5):499-508.
- Edelman SV, Argento NB, Petty SJ, Hirsch IB. Clinical implications of real-time and intermittently scanned continuous glucose monitoring. Diabetes Care. 2018;41:2265-2274.
- Fonseca VA, Grunberger G, Anhalt H, et al. Continuous glucose monitoring: A consensus conference of the American Association of Clinical Endocrinologists and American College of Endocrinology. Endocr Pract. 2016;22(8):1008-21.
- Reiterer F, Polterauer P, Schoemaker M, Schmelzeisen-Redecker G, Freckmann G, Heinemann L, Del Re L. Significance and Reliability of MARD for the Accuracy of CGM Systems. J Diabetes Sci Technol. 2017 Jan;11(1):59-67. doi: 10.1177/1932296816662047. Epub 2016 Sep 25. PMID: 27566735; PMCID: PMC5375072.
- Food and Drug Administration. Premarket Notification 510(k). 2022. Accessed February 25, 2023. https://www.fda.gov/medical-devices/premarket-submissions-selecting-and-preparing-correct-submission/premarket-notification-510k.
- Isaacs, Diana. The pharmacist’s role in continuous glucose monitoring. Pharmacy Today. 2020;26:37-54.
- Battelino T, Danne T, Bergenstal RM, et al. Clinical Targets for Continuous Glucose Monitoring Data Interpretation: Recommendations from the International Consensus on Time in Range. Diabetes Care. 2019;42(8):1593-1603.
Direct download: 163-cgm.mp3
Category: general
-- posted at: 7:00am EDT
|


